Draw the structures of [Co(NH3)6]^3+, [Ni(CN)4]^2- and [Ni(CO)4]. - Sarthaks eConnect | Largest Online Education Community
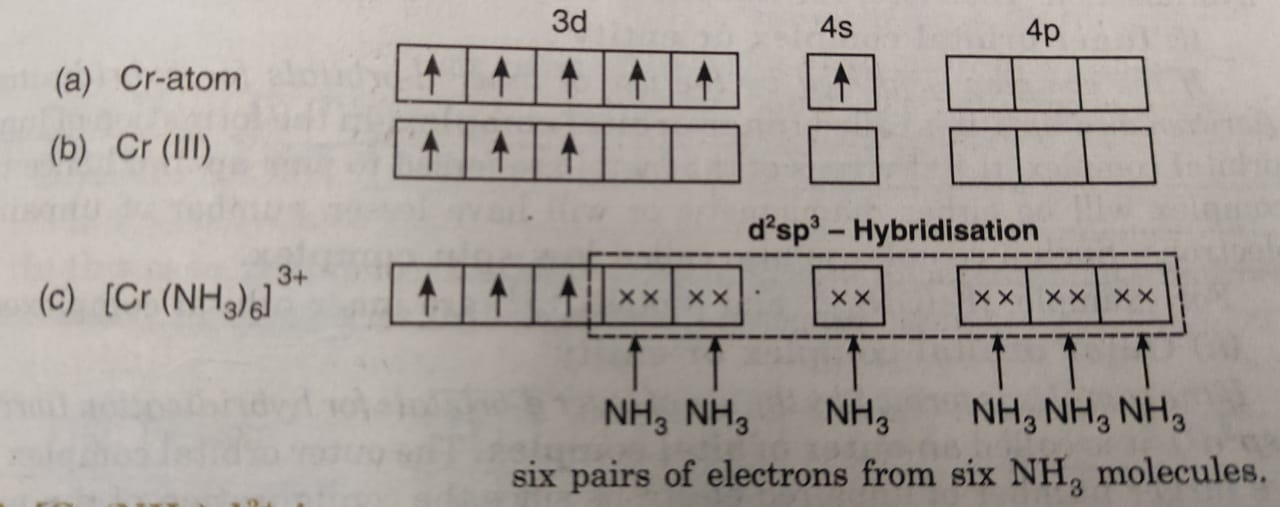
Valence Bond Theory For Bonding In Coordination Compounds - Chemistry, Class 12, Coordination Compounds
![Explain on the basis of valence bond theory that [Ni(CN) 4 ] 2- ion with square planar structure is diamagnetic and [Ni(Cl) 4 ] 2- the ion with tetrahedral geometry is paramagnetic. Explain on the basis of valence bond theory that [Ni(CN) 4 ] 2- ion with square planar structure is diamagnetic and [Ni(Cl) 4 ] 2- the ion with tetrahedral geometry is paramagnetic.](https://static.insightsonindia.in/ncertusercontent/solutions/?domain=gF&l=PROJ12136/1516447284424335.png)
Explain on the basis of valence bond theory that [Ni(CN) 4 ] 2- ion with square planar structure is diamagnetic and [Ni(Cl) 4 ] 2- the ion with tetrahedral geometry is paramagnetic.
![Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fill:224:224/0*8MKpf7WYyO4EenzP.jpg)
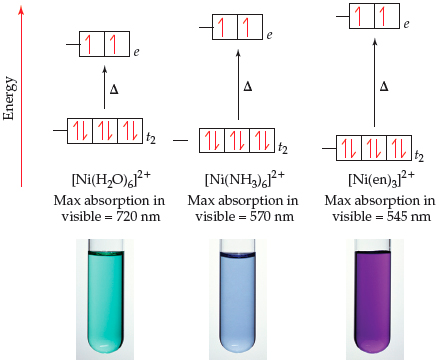
Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
![Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fit:598/0*gC7TqoG4Szy6QwnB.jpg)
Why is [Ni (NH3)6]2+paramagnetic while [Ni (CN) 6]4-diamagnetic? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
Explain on the basis of valence bond theory that [Ni(CN)4]2− ion with square - Sarthaks eConnect | Largest Online Education Community
![Using valency bond approach predict the shape and magnetism (paramagnetic or diamagnetic) of [Ni(CN)4]–. from Chemistry Coordination Compounds Class 12 Manipur Board Using valency bond approach predict the shape and magnetism (paramagnetic or diamagnetic) of [Ni(CN)4]–. from Chemistry Coordination Compounds Class 12 Manipur Board](https://www.zigya.com/application/uploads/images/chen12070392_57148f3062950.png)
Using valency bond approach predict the shape and magnetism (paramagnetic or diamagnetic) of [Ni(CN)4]–. from Chemistry Coordination Compounds Class 12 Manipur Board
![Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson](https://i.pinimg.com/originals/a2/20/10/a22010863c43298057a81f0eb09dd692.jpg)
Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson
![Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ]. Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ].](https://haygot.s3.amazonaws.com/questions/874625_947137_ans_e945a0328151431c8601af56eaa7dcb3.png)
Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ].
![what is the geometry of [Ni(CN)6]4-,,,can i have its structure ,,orbital structure - Chemistry - Coordination Compounds - 6885308 | Meritnation.com what is the geometry of [Ni(CN)6]4-,,,can i have its structure ,,orbital structure - Chemistry - Coordination Compounds - 6885308 | Meritnation.com](https://img-nm.mnimgs.com/img/study_content/content_ck_images/images/Ni%20oh.png)
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-2.png)

![Cr(NH_3)_6]^(3+)` is paramagnetic while `[Ni(CN)_4]^(2-)` is diamagnetic. Explain why?... - YouTube Cr(NH_3)_6]^(3+)` is paramagnetic while `[Ni(CN)_4]^(2-)` is diamagnetic. Explain why?... - YouTube](https://i.ytimg.com/vi/2ukVHaMEd1I/maxresdefault.jpg)
![What is the hybridization of [Ni(CN) 4]-2? - Quora What is the hybridization of [Ni(CN) 4]-2? - Quora](https://qph.cf2.quoracdn.net/main-qimg-20bb619759136ec1b4419262dd5a28a0.webp)
![Write the hybridization and shape of the following complexe : [Ni(CN)4]2– - Chemistry | Shaalaa.com Write the hybridization and shape of the following complexe : [Ni(CN)4]2– - Chemistry | Shaalaa.com](https://www.shaalaa.com/images/_4:ffa0132b7d904ae4be8cc8b622fc0250.png)
![coordination compounds - Electronic configuration in [Ni(CN)4]2- - Chemistry Stack Exchange coordination compounds - Electronic configuration in [Ni(CN)4]2- - Chemistry Stack Exchange](https://i.stack.imgur.com/znHnV.png)
![Hybridization and geometry of [Ni(CN)4]^2 - are: Hybridization and geometry of [Ni(CN)4]^2 - are:](https://haygot.s3.amazonaws.com/questions/1453910_739662_ans_1b88a50e6b3142878d714df46d9472b7.png)
![Ni(CO)4, [Ni(CN)4]2-, [NiCl4]2-Structure-Hybridization-VBT-IIT JEE NEET SAT NCERT CBSE - YouTube Ni(CO)4, [Ni(CN)4]2-, [NiCl4]2-Structure-Hybridization-VBT-IIT JEE NEET SAT NCERT CBSE - YouTube](https://i.ytimg.com/vi/r_C4yyTUSjM/mqdefault.jpg)