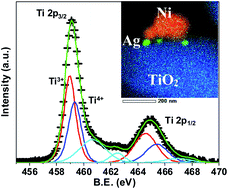
Synergistic effect of Ni–Ag–rutile TiO2 ternary nanocomposite for efficient visible-light-driven photocatalytic activity - RSC Advances (RSC Publishing)
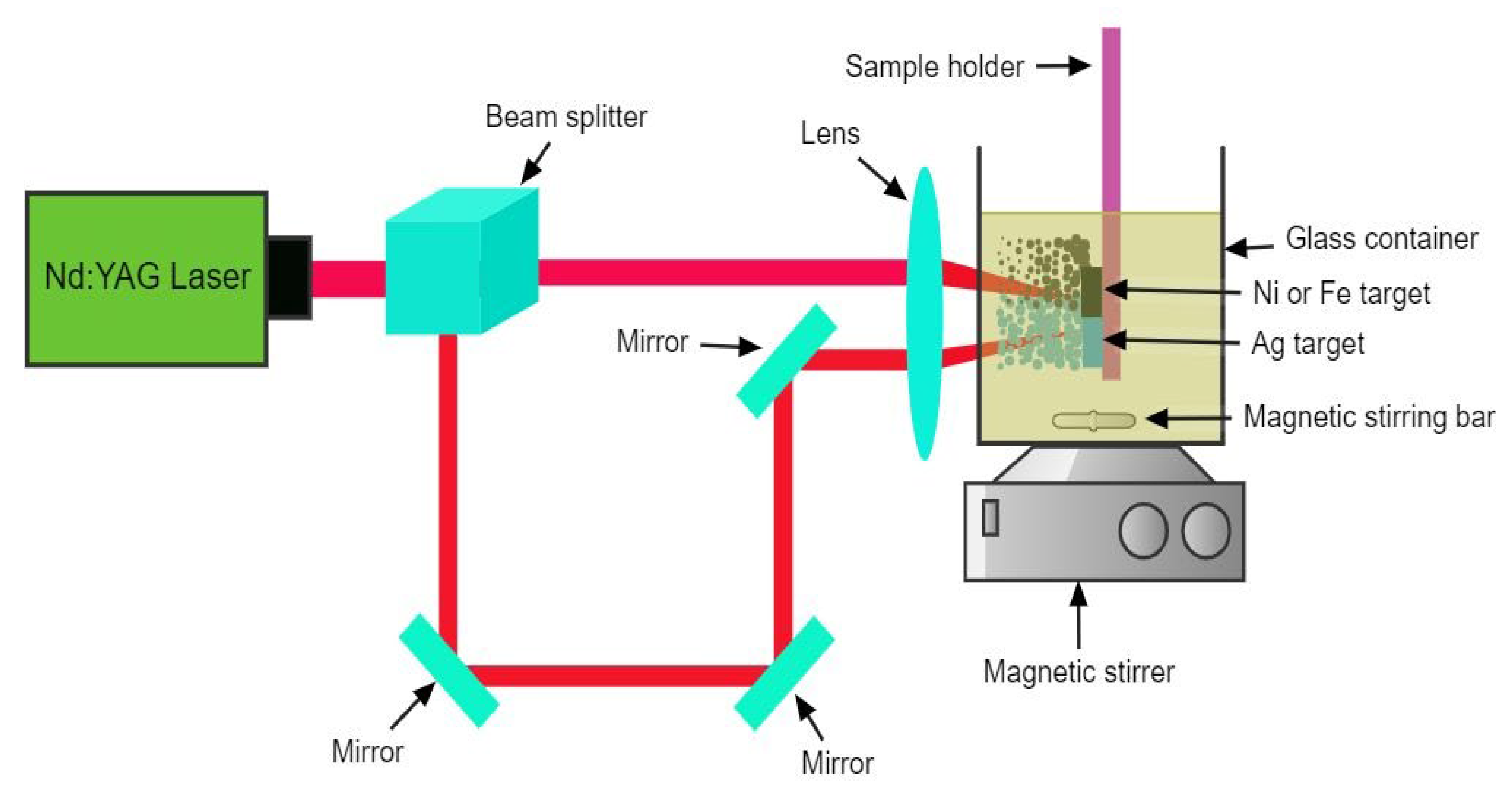
Magnetochemistry | Free Full-Text | Photothermal Hyperthermia Study of Ag/Ni and Ag/Fe Plasmonic Particles Synthesized Using Dual-Pulsed Laser

Overcoming Limitations in Decarboxylative Arylation via Ag–Ni Electrocatalysis | Journal of the American Chemical Society
![PDF] Electrodeposition Approaches to Deposit the Single-Phase Solid Solution of Ag-Ni Alloy | Semantic Scholar PDF] Electrodeposition Approaches to Deposit the Single-Phase Solid Solution of Ag-Ni Alloy | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c1ba0da79d677fe2eaf76e8524e5a19de7e6ebf0/3-Figure1-1.png)
PDF] Electrodeposition Approaches to Deposit the Single-Phase Solid Solution of Ag-Ni Alloy | Semantic Scholar

XRD patterns of as-prepared samples. X-ray diffraction patterns of a Ag... | Download Scientific Diagram

Synthesis and controlled morphology of Ni@Ag core shell nanowires with excellent catalytic efficiency and recyclability - IOPscience
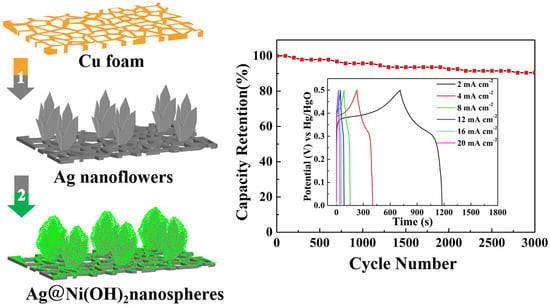
Crystals | Free Full-Text | Hierarchical Core/Shell Structured Ag@Ni(OH)2 Nanospheres as Binder-Free Electrodes for High Performance Supercapacitors
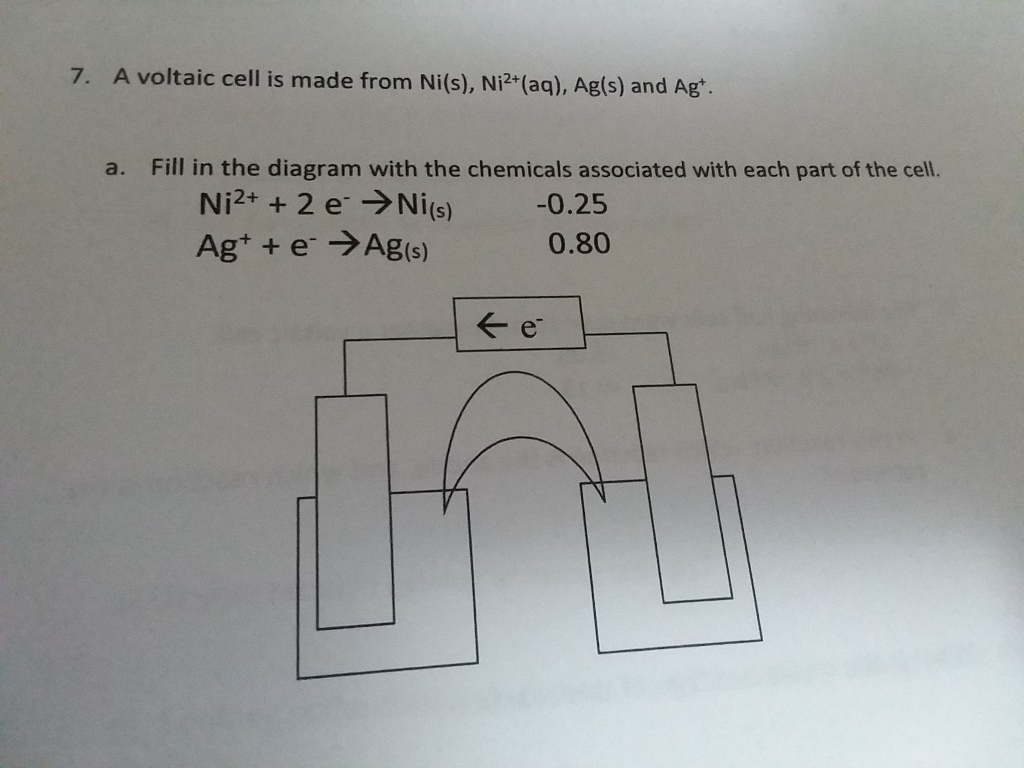
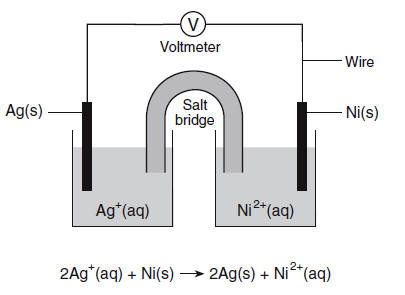
![The Nernst equation the following electrochemical cell will be: Ni(s) | Ni2+ (aq)|| Ag+ (aq)| Ag A) Ecell = Eºcell-RT/F[In[Ni2+]/[Ag+12] B) Ecell = Eccl1-RT/2F[In[Ni2+1/[Ag+1?] C) Ecell = Eºcell-RT/2F[In[Ag+]2/[Ni2+]] D) Ece = Eccl1-RT/2F[In[Ni2+1/[Ag+l] The Nernst equation the following electrochemical cell will be: Ni(s) | Ni2+ (aq)|| Ag+ (aq)| Ag A) Ecell = Eºcell-RT/F[In[Ni2+]/[Ag+12] B) Ecell = Eccl1-RT/2F[In[Ni2+1/[Ag+1?] C) Ecell = Eºcell-RT/2F[In[Ag+]2/[Ni2+]] D) Ece = Eccl1-RT/2F[In[Ni2+1/[Ag+l]](https://toppr-doubts-media.s3.amazonaws.com/images/7675793/5d881151-4f95-4d83-905f-b38d5038d89e.jpg)
The Nernst equation the following electrochemical cell will be: Ni(s) | Ni2+ (aq)|| Ag+ (aq)| Ag A) Ecell = Eºcell-RT/F[In[Ni2+]/[Ag+12] B) Ecell = Eccl1-RT/2F[In[Ni2+1/[Ag+1?] C) Ecell = Eºcell-RT/2F[In[Ag+]2/[Ni2+]] D) Ece = Eccl1-RT/2F[In[Ni2+1/[Ag+l]

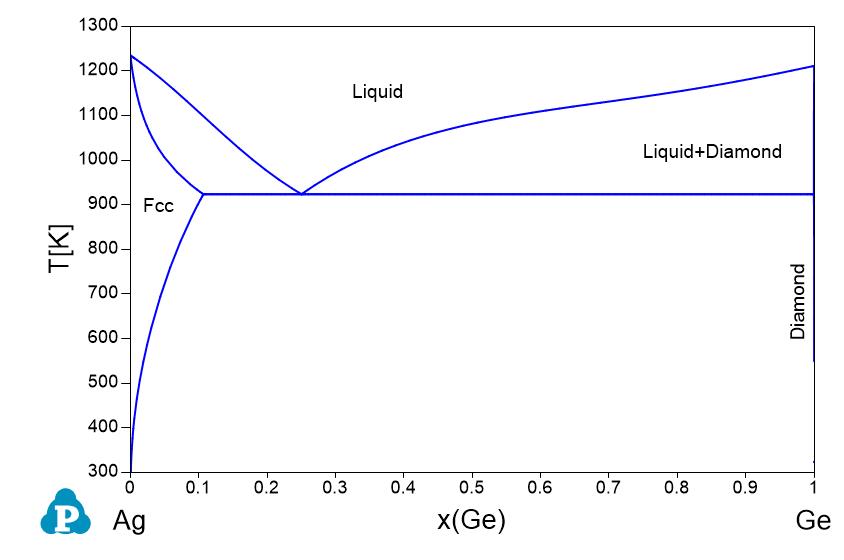
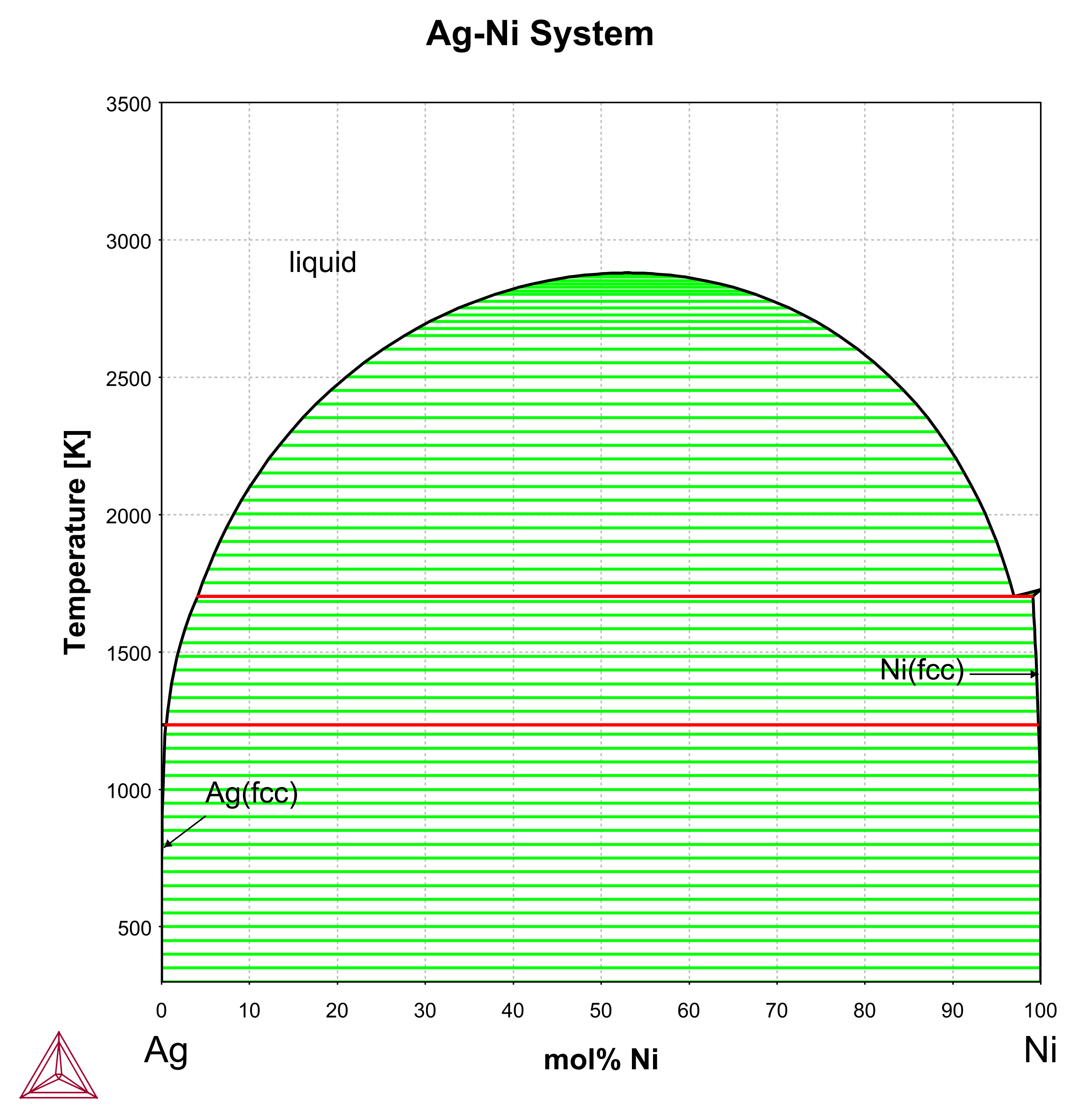
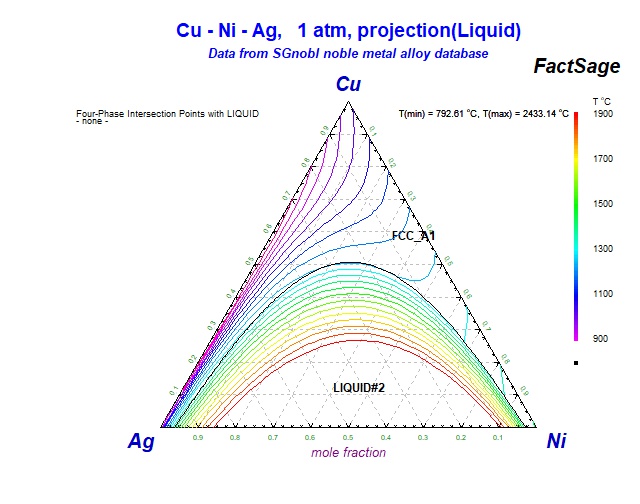
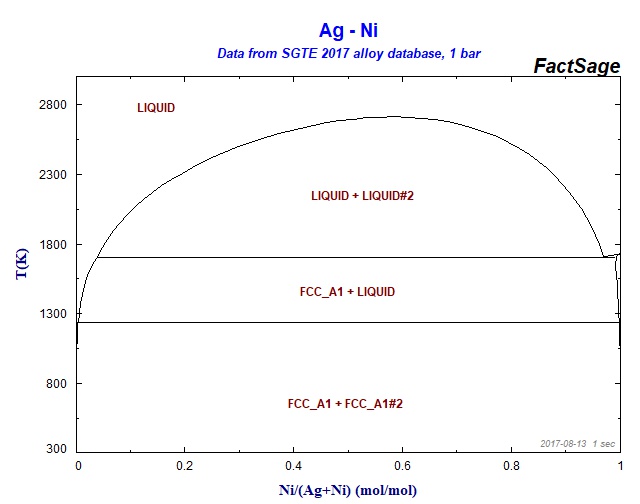
Supplemental Literature Review of Binary Phase Diagrams: Ag-Ni, Ag-Zr, Au-Bi, B-Ni, Co-Sb, Cu-Mn, Cu-Si, Cu-Zn, Fe-Zr, Li-Sb, Mg-Pu, and Si-Zr | Journal of Phase Equilibria and Diffusion

Electrochemical characteristics of silver/nickel oxide (Ag/Ni) for direct ammonia oxidation and nitrogen selectivity in paired electrode system - ScienceDirect